Is plastic cleistogamy an adaptive reproductive strategy?
Cleistogamy is the ability of some plants to produce closed, obligatorily selfed flowers (cleistogamous flowers, CL). Most cleistogamous species also produce open, potentially outcrossed flowers (chasmogamous, CH). The proportion of CL flowers is often plastic and can vary in response to environmental cues. Cleistogamous plants offer the unique possibility to address the evolutionary enigma of the maintenance of mixed-mating systems (Oakley et al. 2007). Namely, mixed mating cannot be maintained simply based on the direct fitness advantages of each mating type (i.e. automatic advantage of selfing and inbreeding depression avoidance through outcrossing), and other factors need to be in play for mixed-mating systems to be evolutionarily stable (Lloyd 1979). Beyond the effects of mixed mating, CL flowers have reduced size, lack reward structures, and pollinate autonomously. It is therefore expected CL flowers to be favored in resource-poor environments that restrict plant growth regardless of the fitness advantage of outcrossing, and in environments with scarce pollinators where they provide reproductive assurance (Kalisz et al. 2004; Heywood and Smith 2021). The fitness advantage of each floral type would be optimal if the plant could reliably predict environmental variation and adjust its mating system accordingly (Lloyd 1992). I studied the complex effects of biotic and abiotic environmental variation on the expression of plastic cleistogamy in an annual ruderal species, Lamium amplexicaule. I notably examined how cleistogamy varied in populations with contrasting ecological characteristics, and whether the observed variation is adaptive. I investigated several possible avenues to explain the ecological factors that govern the expression of cleistogamy and its adaptive function.
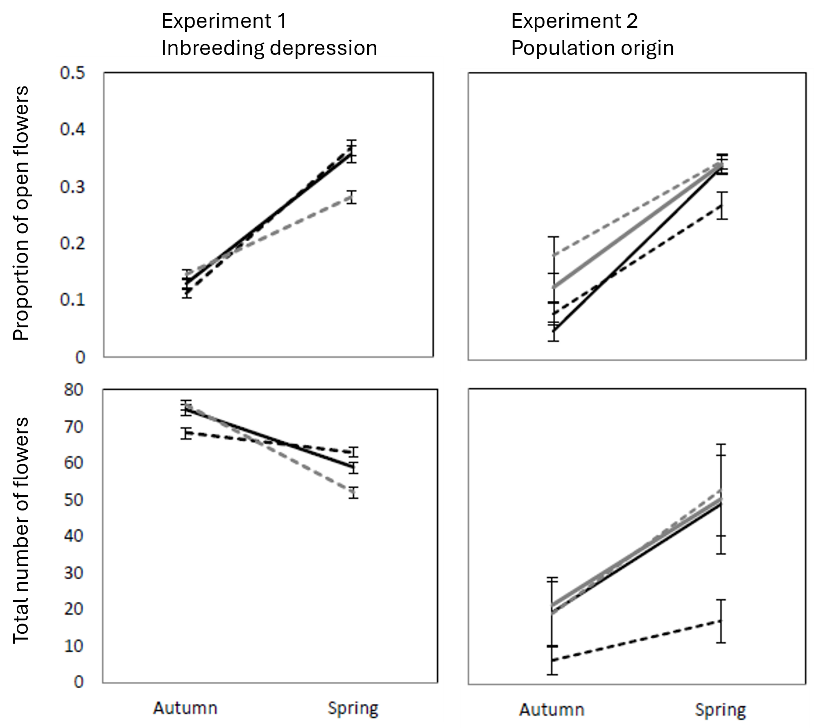
Through a series of field observations and experimental studies, I showed that the proportion of closed, cleistogamous flowers varies in response to a range of environmental factors and reliable cues – seasonal variation, photoperiod, drought, intraspecific competition. Regardless of the cue, the proportion of cleistogamous flowers does not depend on the size of the plant, meaning it is not resource limited (Figure 4). Instead, there are likely endogenous factors triggered by reliable environmental cues that forecast the competitive and pollination environment which favor the most suited mating strategy. A lecture presenting some of the key results is available online.
Effects of light competition on mating strategy and transcriptome variation in a cleistogamous plant
I am currently running a controlled environment experiment to test the effects of a specific light cue reliably associated with competition, the Red:Far Red light ratio (R:FR). Since R:FR simultaneously elicits responses specific to competition (e.g. competition avoidance and tolerance, sensu Gruntman et al. (2017)), and an adjustment of the mating strategy (i.e. production of open and closed flowers), I will also use a transcriptomic approach to investigate differential gene expression to identify upstream regulation factors that trigger the expression of cleistogamy before floral initiation.
-
Gruntman, Michal, Dorothee Groß, Maria Májeková, and Katja Tielbörger. 2017. “Decision-Making in Plants under Competition.” Nature Communications 8 (1): 2235. https://doi.org/10.1038/s41467-017-02147-2.
-
Heywood, John S., and Stephanie A. Smith. 2021. “Does Cleistogamous Self-Fertilization Provide Reproductive Assurance against Seed Predation in Ruellia Humilis?” The Southwestern Naturalist 65 (2): 116–22. https://doi.org/10.1894/0038-4909-65.2.116.
-
Kalisz, S., D. W. Vogler, and K. M. Hanley. 2004. “Context-Dependent Autonomous Self-Fertilization Yields Reproductive Assurance and Mixed Mating.” Nature 430 (7002): 884–87. https://doi.org/10.1038/nature02776.
-
Lloyd, David G. 1979. “Some Reproductive Factors Affecting the Selection of Self-Fertilization in Plants.” American Naturalist 113 (1): 67–79.
-
Lloyd, David G. 1992. “Self-and Cross-Fertilization in Plants. II. The Selection of Self-Fertilization.” International Journal of Plant Sciences 153 (3, Part 1): 370–80.
-
Oakley, Christopher G., Ken S. Moriuchi, and Alice A. Winn. 2007. “The Maintenance of Outcrossing in Predominantly Selfing Species: Ideas and Evidence from Cleistogamous Species.” Annual Review of Ecology, Evolution, and Systematics 38: 437–57. https://doi.org/10.1146/annurev.ecolsys.38.091206.095654.